Combating Metastases
What is Combating Metastases?
Metastatic cancer is the term used to describe any cancer that has spread from the area it started in to other areas of the body. For some types of cancer, metastasis is also called stage IV cancer, and metastatic cancers are often hard to control.
Cancer can spread to any part of the body, but the most common sites of metastasis are the bones, brain, liver and lungs. Although metastasis causes more than 90% of cancer-related deaths, it only receives less than 5% of research funding.
Support Breakthroughs in Combating Metastases Research and Beyond.
Related Content
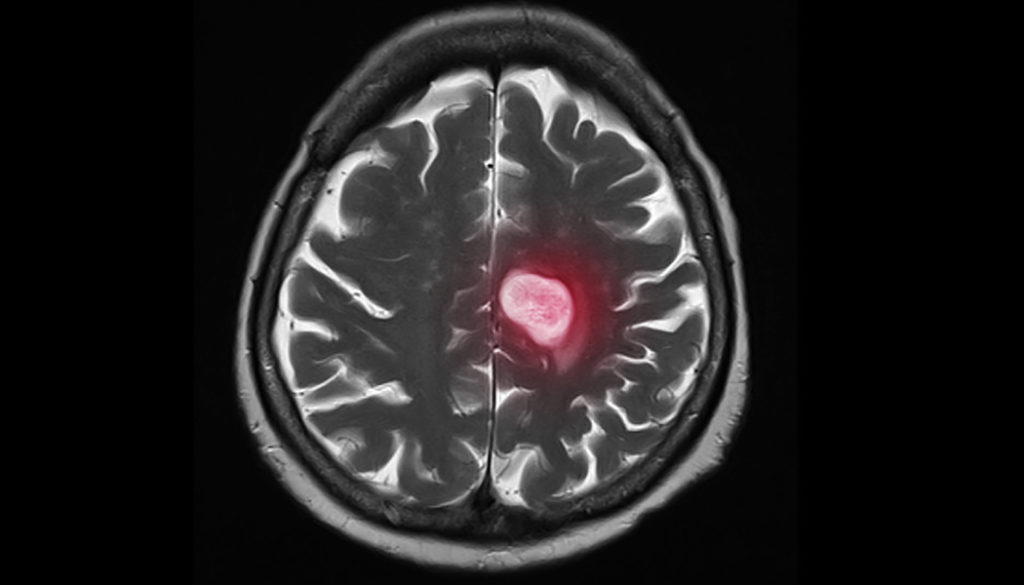
Research Highlight: Preventing Breast Cancer Brain Metastasis
National Foundation for Cancer Research funded researcher Dr. Daniel A Haber recently unearthed an exciting discovery that may add years to the lives of late-stage breast cancer patients. Dr. Haber is fascinated by understanding drug resistance on a deeper level by studying individual tumor cells in patients’ blood. In December 2020, Dr. Haber and his team shared their exciting findings on how brain metastasis, or the spread of cancer to the brain, may be prevented. Brain metastases occur in about 10% of all patients with cancer and in as many as a third of women with advanced metastatic breast cancer. Though experts have made great strides in suppressing the spread of cancer, there is still little known about the cellular pathways that enable cancer cells to selectively grow in the brain; that is, until Dr. Haber and his research team identified a signaling pathway which appeared significantly more active in brain metastases from breast cancer. “We were looking for what properties of some breast cancer cells made it possible for the cells to grow in the brain, which is a rare but often deadly complication of breast cancer,” Dr. Haber explained. “We weren’t sure what we would find. In a way that’s what makes the discovery process so exciting.” The research commenced approximately 10 years ago while investigating circulating tumor cells (CTCs). As their research progressed, the team homed in on a specific signaling pathway named HIF1A. Using cells from women with breast cancer, the team observed how these cells acted in animal models. It was discovered that if HIF1A was suppressed, the rate of proliferation (or rapid growth) was reduced. Simply put, blocking the HIF1A signaling pathway could reduce the rate of brain metastasis or even prevent it all together. “HIF1A is not specific to brain metastasis, since it’s a very broadly activated pathway in many cancers,” Dr. Haber began. “However, it appears to be more active in brain metastases from breast cancer than in primary breast cancers, and that may help explain what makes these metastases to the brain so unique and so difficult to treat.” The pathway identified in this research is already well known in the cancer world, however its special relevance to the brain was not known until now. Theoretically, a drug could be developed to suppress HIF1A and, in turn, prevent the spread of cancer. While a very promising discovery, Dr. Haber explains that there is far more work to be done. “There are a few HIF1A suppressing drugs now being tested in clinical trials for other indications,” Dr. Haber said, “However, we would have to expand this to multiple different models and systems before we could contemplate an intervention.” In regard to the length of time it takes to have a discovery such as this transitioned to mainstream treatment, Dr. Haber explains that it “depends on the discovery, its potential applications and some ‘luck’.” While the process from discovery to clinical treatment is getting faster all the time, it is likely going to take five to ten years before this finding is implemented into treatment. As for Dr. Haber, he has already planned plenty of work […]
The Hallmarks of Metastasis
It is the one word you do not want to hear in a cancer diagnosis: metastasis. It represents the figurative death-knell; once a cancer has spread to other parts of the body, survival rates plummet and what treatments do exist switch from curative to palliative. The dividing line metastatic cancer (stage IV cancer) can even be seen in funding, where 93 percent of monies go to preventative measures and early detection, rather than for therapies to combat cancer on the move within the body. And yet, in understanding the mechanisms of metastasis, the cure for this stage of cancer may be found. It is the reason investigators are taking a fresh look at exactly what happens when cancerous cells first break away from their host tumor. Not surprisingly, metastasis is a complex biological process; it is actually an amalgam of several different events. Douglas Hurst, Ph.D., assistant professor in the UAB Department of Pathology, and Danny Welch, Ph.D., associate director of Education at the KUCC and NFCR funded scientist, conducted a literature review of more than 10,000 publications on metastasis, and found that in order for a cancerous cell to be “metastatic,” a cell has to exhibit the following qualities: Motility and Invasion By definition, metastatic cells must be able to migrate from the original tumor. Invasion, the defining feature of malignancy, is the capacity for tumor cells to disrupt the basement membrane (a thin, fibrous, extracellular matrix of tissue that separates the lining of an internal or external body surface from underlying connective tissue) and penetrate underlying stroma (the tumor’s support structure.). Motility alone is not sufficient; if a cell successfully breaks away from a tumor but subsequently dies, it is not considered metastatic. Ability to Modulate the Secondary Site Metastatic cancer’s ability to co-opt local tissue is a wonder of biology. In one fell swoop, cancer recruits new cells into the local microenvironment, elicits mobilization of immune/inflammatory cells, restructures other tissues, alters metabolism of surrounding stroma, cancels any antitumor actions by the immune system, manipulates the behavior of other cancer cells, alters the extracellular matrix, and restructures normal behaviors of other cells. Plasticity Primary or secondary, tumors are not static; on a cellular level they are, in fact, very dynamic. Cancer cells must adapt their metabolism to meet the demands that accompany rapid growth of the primary tumor and colonization of distinct metastatic sites. Moreover, neoplastic cells can alter growth rates of other cells, drug resistance, and metastatic capability. Ability to Colonize Secondary Tissues Colonization of secondary tissues is the standout feature of metastatic cancer. In the Hurst-Welch study, it is suggested that if a tumor cell or cells breaks away from the primary tumor and survives, but cannot take root elsewhere in the body, metastasis is not taking place. Once metastasis occurs, survival rates can plummet as much as 90 percent, so it is critical to discern the events of the entire metastatic cascade. Recent research shows promise: Welch, an NFCR-funded fellow, and his team have discovered eight of the 35 known cancer metastasis suppressor genes. Further study based on these discoveries may lead to the design […]
Liquid Biopsy Advances: Capturing Metastatic Cancer Cells
A novel instrument for detecting cancer cells in the blood could lead to individualized cancer treatments for patients suffering metastasis. Developed by a Massachusetts General Hospital team led by Daniel Haber, M.D., Ph.D., a renowned cancer scientists supported by the National Foundation for Cancer Research since 2003, the business card-sized CTC-iChip captures circulating tumor cells—referenced in the name of the device— “simply” by running blood from a cancer patient across its surface. Studded with microposts coated, or “functionalized,” with antibodies designed to seize CTCs in mid-flow, the captured cells can not only be confirmed as tumor-related, but also counted and further analyzed in a variety of ways including molecular characterization. From there, clinicians can decide on the proper treatment or develop new ones. Genetic testing on captured CTCs may give doctors a way to more effectively treat tumors or stop them from spreading. “Tumors evolve in response to treatment,” said Haber. “They often acquire new genetic features that make them resistant to drugs. If we can monitor those changes in real time, we stand a better chance of matching the right treatments with the right patients against those new changes in their tumors.” Circulating tumor cells are tumor cells that have become detached from the primary tumor and enter blood circulation. While CTCs occur once in a billion cells, and therefore are extremely rare, they nevertheless may hold a key to metastasis—the stage responsible for the vast majority of cancer deaths. While still in trial stages, the CTC-iChip could prove to be a powerful tool in managing metastatic cancer care on several fronts. Because it is utilized via a simple blood sample, the need for biopsies and their associated pain and healing period is eliminated. Moreover, CTC monitoring on the spot allows the response of any applied treatments to be monitored in real time, as well as any resistance to them. Additionally, the CTC-iChip captures circulating tumor cells when they are still viable—dead cells are not useful for long-term analysis. The device could dramatically improve treatment and diagnosis for many different types of metastatic cancers, including metastatic breast cancer and glioblastoma. The CTC-iChip is already responsible for some fundamental discoveries in cancer research. Through experimentation in the lab, the researchers have found a unique and dynamic interconversion of HER2 gene expression: ER+/HER2- cells could spontaneously become ER+/HER2+ and convert back to ER+/HER2-. Because the HER2 gene expression oscillates between “on” and “off” spontaneously in these tumors, researchers reason that the two separate signaling pathways may need to be simultaneously turned off to halt either cell group from repopulating one another and beginning new abnormal growth. While more research is needed, it is an exciting start to CTC-iChip technology. Dr. Haber and his team are now focused on optimizing CTC culture conditions, improving efficiency, bringing down costs and meeting the remaining challenges to bring individualized therapy into the clinic. And NFCR remains committed to lending its support to these exciting and potentially life-saving efforts. References: http://cancerdiscovery.aacrjournals.org/content/early/2018/08/12/2159-8290.CD-18-0432 https://www.jto.org/article/S1556-0864(15)30978-3/fulltext https://www.massgeneral.org/cancerresearch/research/researchlab.aspx?id=1171 https://www.nfcr.org/scientists/daniel-haber/ http://www.prweb.com/releases/2014/07/prweb12042342.htm https://www.sciencedaily.com/releases/2016/09/160906213434.htm http://science.sciencemag.org/content/345/6193/216/tab-figures-data